World Cup's 14-mile Green Corridor to leave lasting impact on Houston
Big Win
The FIFA World Cup 2026 Houston Host Committee has announced new details about its massive Green Corridor project, including the many improvements that will outlast the iconic sporting event taking place in Houston this summer.
The Green Corridor will be a 14-mile long verdant artery connecting multiple major landmarks in Houston through safe, walkable paths that include shade trees and other improvements. First conceived in 2024 by the Sustainability Subcommittee led by Elizabeth Carlson, it will unite East Downtown, Downtown, Midtown, the Museum District, and Third Ward through a hike and bike trail as well as METRO Rail stops. Though the Green Corridor is beginning its life as a showcase for the city to visitors attending the FIFA World Cup June 14 -July 4, it will remain a permanent installation for Houstonians to travel the city without cars.
Management of the project is being handled by Impact Houston 26, a portion of the Host Committee empowered by the Harris County-Houston Sports Authority’s Sports Authority Foundation to promote long-term benefits to the city after the World Cup. Funding partners include private corporations as well as civic organizations such as the City of Sugar Land and Rice University.
“The Green Corridor reflects what Impact Houston 26 is all about, using the FIFA World Cup as a catalyst to deliver lasting environmental benefits for our city,” Carlson said in a statement. “Through Impact Houston’s pillar on sustainability, we’re able to collaborate with local stakeholders to create not just demonstrations of resilience and innovation but education and engagement in the community, a meaningful legacy long after 2026.”
The corridor will provide access to both Houston Stadium (also known as NRG Stadium) and the FIFA Fan Festival, as well as improve existing paths like the Columbia Tap Trail in Third Ward. These improvements include the installation of shade structures, native plantings, expanding the tree canopy, air quality monitoring devices, and water and bike repair stations.
Impact Houston 26 is also working with local institutions like the Houston Zoo, Greentown Labs, and Discovery Green to install various educational materials along the Green Corridor.
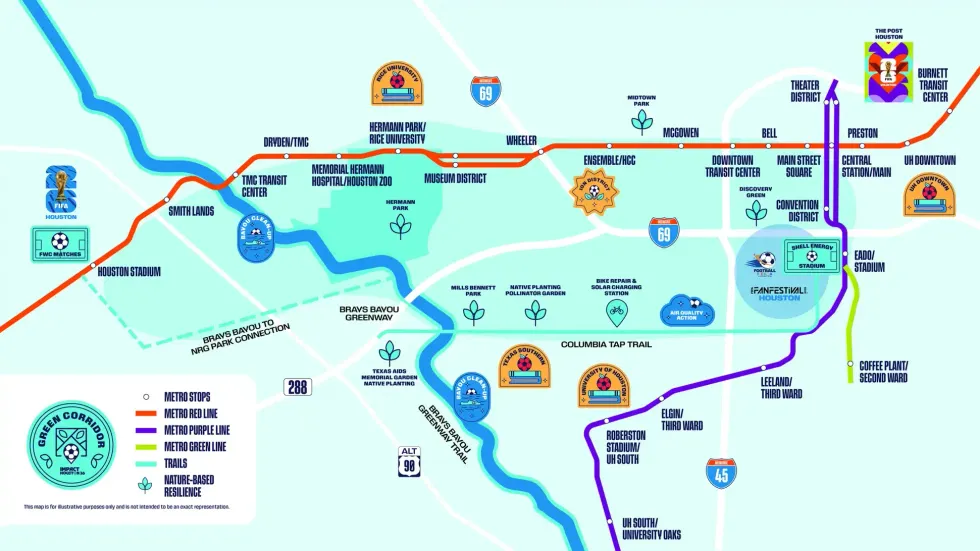
Below is a breakdown of other improvements planned or completed as part of the Green Corridor.
- Downtown Houston Main Street Promenade: Four permanent shade structures, native plants, and expanding the tree canopy by 154% to be implemented by May 2026. Further shade structures and plantings planned for Texas Avenue.
- East Downtown Management District: Native tree plantings and landscaping in and around the FIFA Fan Festival site to improve first/last mile connectivity around the Green Corridor.
- Columbia Tap Trail: Installation of 325 solar lights.
- Stadium Park/Astrodome and TMC/Dryden plus Fannin South Transit Center: Various landscaping and safety enhancements.
- Midtown Houston: $1.5 million in landscaping and beautification along the Red Line, including over 80 trees, native plantings, water stations, waste receptacles, crosswalk improvements, and public art installations.
The Green Corridor is only one of the World Cup Host Committee's sustainability initiatives. In January, it announced the "New Year, New Hou" program that provides hospitality businesses such as restaurants and hotels with one of three certifications.
---
This article originally appeared on CultureMap.com.
